|
9/9/2023 0 Comments Dalton atomic theory (6) Atom is no longer Indestructible: By carrying out nuclear reactions atom of an element may be changed into another. theory that really made a lot of sense in comparison to what we know to be true is John Dalton's atomic theory so in your Chemistry Book. He was the first to suggest that atoms are the main constituents of matter. (5) Atom is the smallest particle that take part in a chemical reaction. Chemist John Dalton developed a theory that devised the concept of atoms. 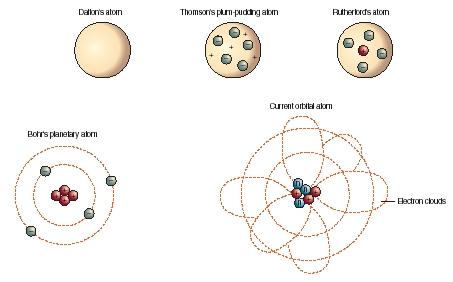
(4) The ratio in which the different atoms combine with one another may be fixed and integral but may not be simple :įor example : In sugarcane the ratio in which the elements carbon, hydrogen and oxygen combine together 12 : 22 : 11 which is not simple. Their speculation about a hard, indivisible fundamental particle of nature was replaced slowly by a scientific theory supported by experiment and. (3) Atoms of different elements may have same atomic masses: Atoms of the different elements which have the same atomic masses are called isobars.įor Example : Calcium and Argon have atomic masses of 40 with atomic number as 20 and 18. The concept of the atom that Western scientists accepted in broad outline from the 1600s until about 1900 originated with Greek philosophers in the 5th century bce. Previously, we defined an atom as the smallest part of an element that maintains the identity of that element. His famous disciple, Democritus of Abdera, named the building blocks of matter atomos, meaning literally indivisible, about 430 bce. 1 ), is a fundamental concept that states that all elements are composed of atoms. The atomic philosophy of the early Greeks Leucippus of Miletus (5th century bce) is thought to have originated the atomic philosophy. (2) Atoms of the same element may have different atomic masses: Atoms of the same element which possess different atomic masses are called isotopes. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 2.2. (1) Atom is no longer considered to be indivisible: It is made up of electrons, protons and neutrons.  /John_Dalton_by_Charles_Turner-56a1338a5f9b58b7d0bcfc9e.jpg)
(3) It could not explain the nature of binding forces between atoms and molecules which accounts for the existence of three States of matter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
